Tag: FDA bioequivalence
Bioequivalent Medications: What the Term Really Means
Bioequivalent medications are generic drugs proven to work the same way as brand-name versions in your body. Learn how regulators test for equivalence, why it matters, and what the data says about real-world use.
Read moreGeneric Absorption Rates: How They Must Match Brand Drugs to Be Safe and Effective
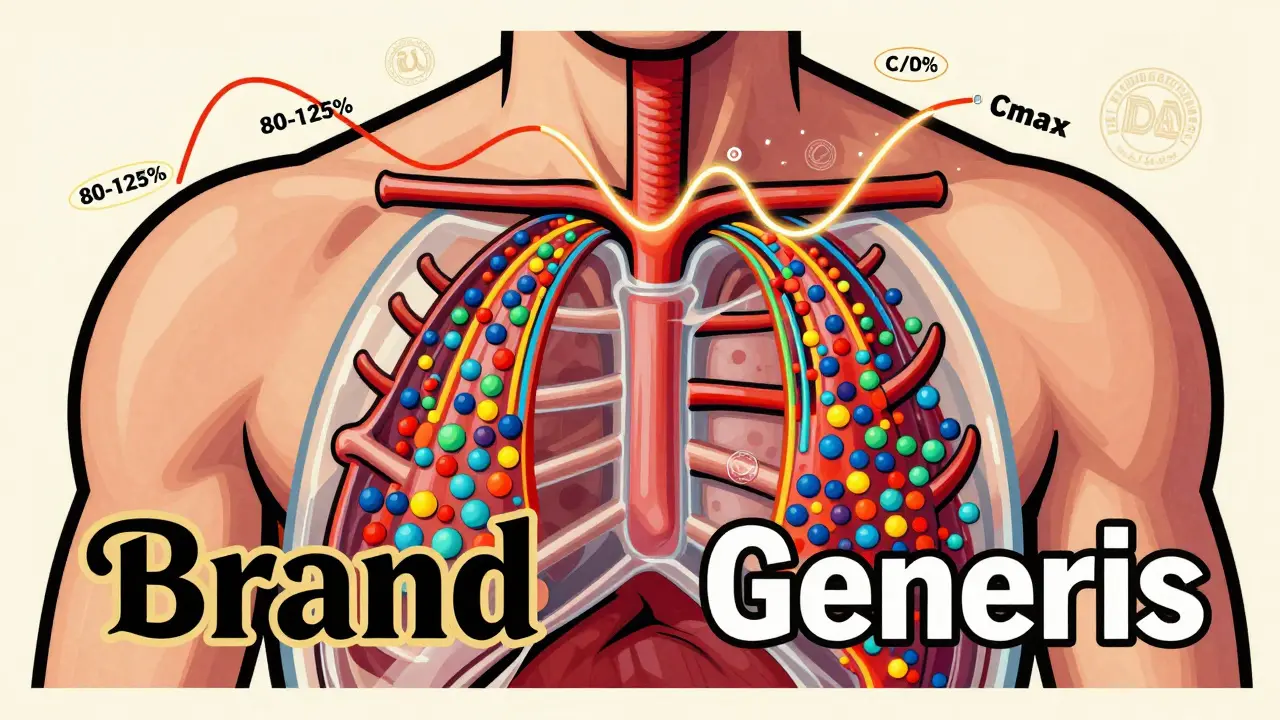
Generic drugs must match brand-name absorption rates within strict FDA limits to be approved. Learn how bioequivalence works, why some patients feel differences, and why generics are safe and effective.
Read moreIn Vivo vs In Vitro Bioequivalence Testing: When Each Is Used
In vivo and in vitro bioequivalence testing are key to approving generic drugs. In vivo uses human trials; in vitro uses lab tests. Learn when each is required and why regulators choose one over the other.
Read more